Earlier this year, CMS announced that HHVBP (Home Health Value Based Purchasing) would be expanded after learning that participants in the HHVBP model had an average of 4.6% improvement in quality scores and an average annual savings of $141 million to Medicare. In a recent federal register rule proposal, CMS proposes including all Medicare-certified home health agencies (HHAs) in an HHVBP Model, starting January 1, 2022 for CY2024 payment, with CY2019 data used to calculate the model’s scoring thresholds. This model has a proposed maximum adjustment of ±5%, with average performance compared to other HHAs in their cohort receiving no adjustment.
The suggested model divides HHAs into two cohorts: larger-volume HHAs that receive 40 or more completed HHCAHPS surveys in a performance year, and smaller-volume HHAs that receive fewer than 40 completed surveys. The original model also divided HHAs by state, however, this is being discontinued in the proposed model to simplify scoring and ensure that all cohorts are large enough to provide valid scores, although the rule also requests comments on an alternative approach of only using state/territory-based cohorts (without volume considerations) instead, which may be finalized after considering comments.
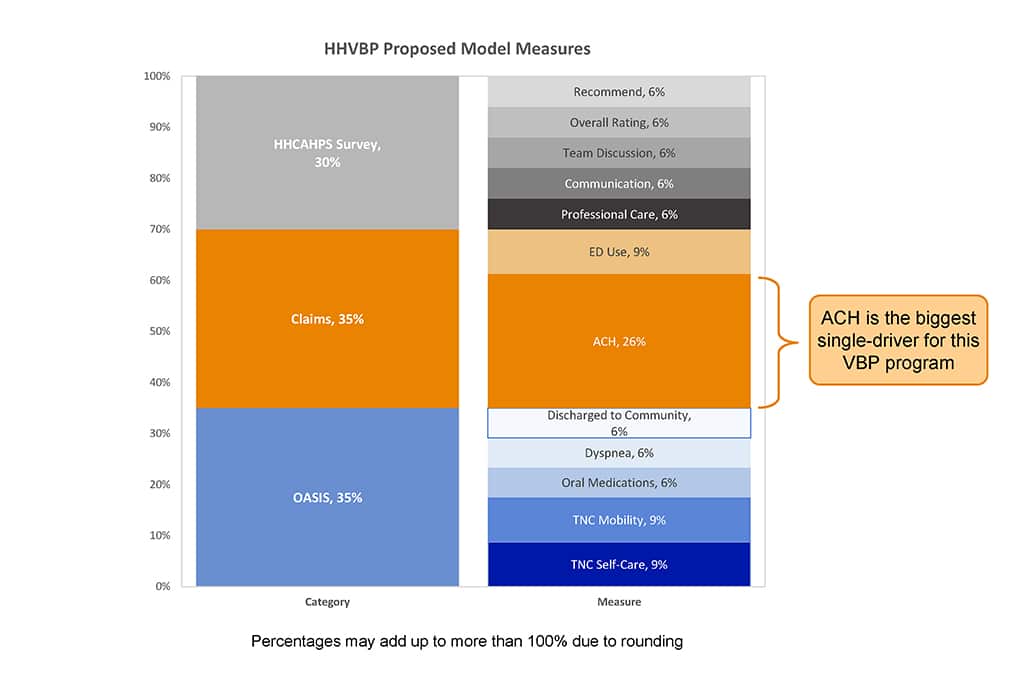
The HHVBP Model, as proposed will use data already reported by HHAs, including measures in three categories: OASIS, Claims, and HHCAHPS survey data. Each of the five HHCAHPS survey measures account for 6% of the Overall HHVBP Total Performance Score for a combined 30%. This includes the Willingness to Recommend item, which was excluded from HCAHPS VBP. OASIS and Claims data each make up 35% of the TPS score, with Acute Care Hospitalization During the First 60 Days of Home Health Use (ACH), as part of the Claims measure, making up more than one-quarter of the entire TPS score.
For each measure, CMS would calculate an achievement threshold and a benchmark for each cohort to award achievement points and improvement points to evaluate each HHA’s performance against their own earlier performance during the baseline period and the performance of the other HHAs within their cohort.
CMS welcomes comments on this proposed rule, which may be submitted via http://www.regulations.gov by following the instructions under the “submit a comment” tab. Comments must be submitted by 5 pm EDT on August 27, 2021.